|
Case Report
Oral rehabilitation in mandibula with implants and nano-biomaterial, using the split-crest technique: A case report
1 Department of Surgery, Faculty of Dentistry Multivix, Espirito Santo, Brazil
2 Faculty of Dentistry, FAESA, University Center, Campus Vitória, Brazil
3 Faculty of Dentistry, UNIFENAS, José do Rosário Vellano University, Campus Minas Gerais, Brazil
4 Department of Implantology, Faculty of Dentistry, State University of Rio de Janeiro, Rio de Janeiro, Brazil
Address correspondence to:
Igor da Silva Brum
Department of Implantology, Faculty of Dentistry, State University of Rio de Janeiro, Rio de Janeiro,
Brazil
Message to Corresponding Author
Article ID: 100081Z12JR2020
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Rosalem JMM, Engel DP, Bôa MP, Maurity TNY, Brum IS. Oral rehabilitation in mandibula with implants and nanobiomaterial, using the split-crest technique: A case report. J Case Rep Images Surg 2020;6:100081Z12JR2020.ABSTRACT
Introduction: There are many ways to rehabilitate the mandible and maxilla with bone defects, such as autogenous bone graft or biomaterials graft, those techniques can be associated with resorbable or non-resorbable screen. The split-crest technique stands out due to its predictability and low morbidity for the patient, compared to other techniques.
Case Report: In this study the patient had a lack of thickness in the left mandibular region, split-crest technique was chosen in order to increase thickness using a nanobiomaterial (Blue Bone, Regener Biomaterial, Curitiba, Brazil). After five months, a new tomography has been made and the use of Morse Cone implants (Avantt, Systhex, Curitiba, Brazil) was selected for the case, furthermore, it has been planned virtually ceramic laminates for the remaining teeth and a ceramic prosthesis on the implants.
Conclusion: All things considered, the split-crest technique is an excellent technique to increase the bone thickness with a lower cost and morbidity for the patient, which can be indicated for cases of oral rehabilitation using dental implants.
Keywords: Biomaterials, Dental implants, Grafts, Mandibular defects, Prosthesis, Rehabilitation, Split-crest
Introduction
When oral rehabilitation in areas with bone defect is necessary, it becomes a complex issue. The bone thickness deficiency is common for edentulous patients, especially when there is alveolar fracture during tooth extraction [1]. When bone loss results from a maxillofacial trauma, dental vertical root fracture, or extensive endodontic periodontal lesions, the effects are even more severe. These factors can result in insufficient vertical and horizontal support for the installation of dental implants and may compromise the options of definitive ceramic prosthesis [2]. Indeed, the increase of bone volume, on bone defect, would enable and improve the prognosis of oral rehabilitation [3].
We can mention several bone reconstruction techniques, including vertical and horizontal volume increase using autogenous bone or biomaterials, coupled with the use of screen that could be made of titanium, alumina, polytetrafluoroethylene (PTFE), bovine, porcine, or synthetic origins of collagen type 1 and also, reconstructions with prototyping [4]. However, the evolution of dentistry allows us to study different ways of solving most of the complicated surgical situations [5].
da Silva Brum et al. [6] describe the use of nanobiomaterials as an excellent alternative in order to solve the lack of bone thickness for dental implant’s installation. Due to the osteoconductive and osteoinductive characteristics similarity between the nanobiomaterials and the autogenous bone, the variety of material alternative becomes possible, which in many cases is the major problem of autogenous bone technique, considering the insufficiency of donor area [7].
Moreover these surgical techniques for horizontal reconstructions, split-crest, well described in the literature, is also a valuable option for bone reconstruction [8], enabling immediate or late dental implants installation, with or without the use of biomaterials, which improves regeneration between fractured cortices, working as a framework [9].
Systematic reviews and meta-analyzes have documented that the horizontal reconstruction of alveolar defects with split-crest technique appears to be predictable and efficient, due to the high rate of implant survival, the increase of horizontal bone volume allowing the installation of implants, as well as not having negative biological factors and almost no complications when the surgical technique is well performed [10],[11]. As more than 10 years of literature passed by, other variations of the techniques have emerged, making it possible to divide the crest and install immediate implants, being able to use biomaterial or not, with a fixing screw or not. This mix of possibilities makes this technique a viable alternative for cases of oral rehabilitation using dental implants, where the lack of remaining bone is an unfavorable factor [12].
The aim of this case report is to describe the split-crest technique in a left mandibular area, which the lack of remaining bone would have made the dental implants installation, in an ideal position, impossible.
Case Report
Patient
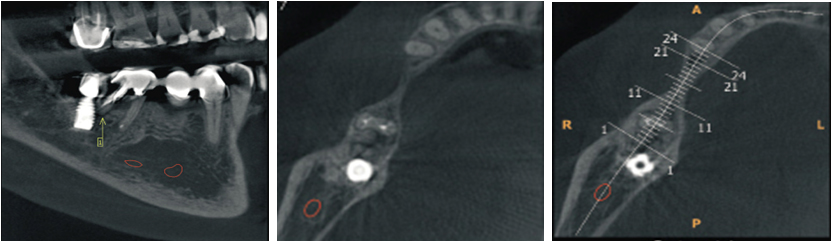
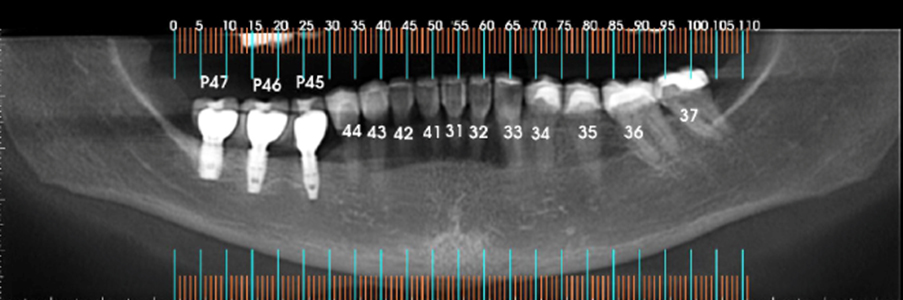
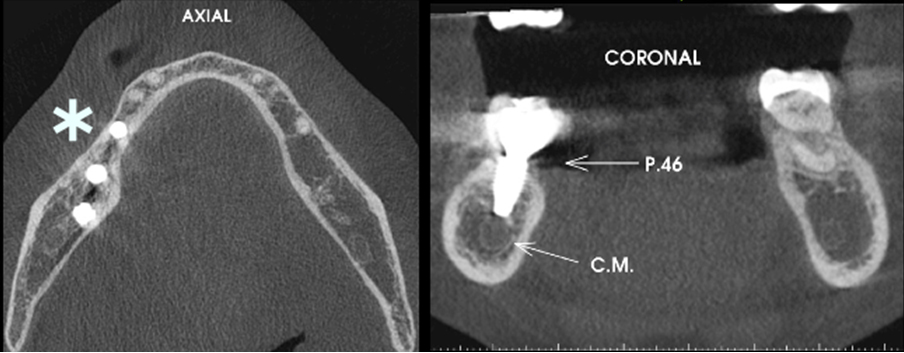
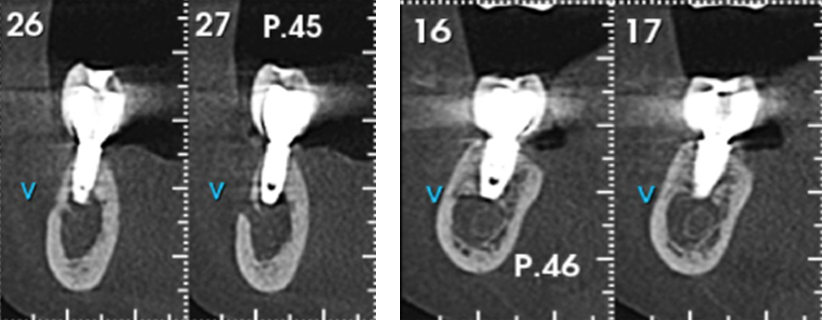
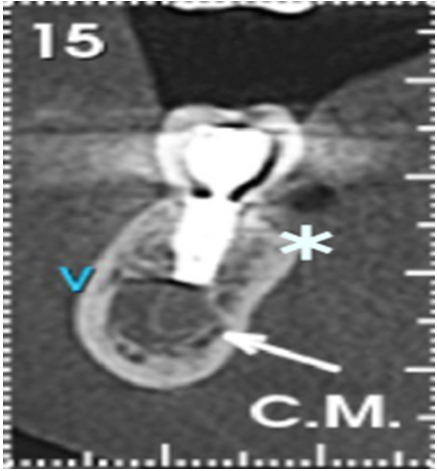
This clinical report followed up the rehabilitation of a 31-year-old female, with leukoderma, who needed an oral rehabilitation with biomaterial and dental implants. Due to the periodontal lesion of the element 46, she had her implant, present in the area of the element 47, compromised (Figure 1). The region of the 45-element was not thick enough in order to install the implant in an ideal position (Figure 2). Therefore, it was planned to remove the element 46, a vigorous curettage of the mesial bone wall of the implant and perform the split-crest technique on the element 45 area, combining with the use of biomaterial, for the bone remodeling process. Dental implant installation was programmed after five months of the grafting process, and then, after four months of the implant installation, we have planned scanned prostheses on the implant and porcelain veneers on the other teeth. The follow-up time from the beginning to the end of the treatment was one year and six months.
Biomaterial and implants
During the reconstruction surgery, a Blue Bone alloplastic NanoBiomaterial from Regener biomaterials, Curitiba, Brazil was used coupled with HeliCote Integra Miltex, USA collagen screen. The two implants used are from Systhex company, model cone morse (Avantt), Curitiba, Brazil, both with the same length and diameter specification (4.3 × 8.5 mm).
Operative and post-operative and prosthetic manipulation
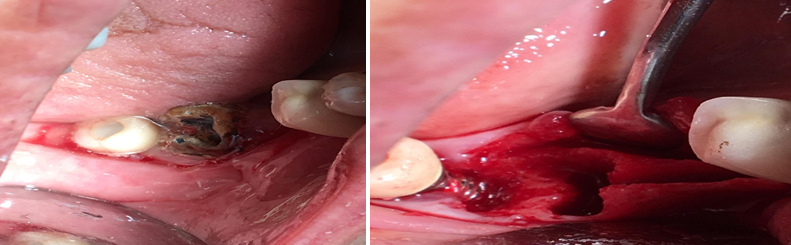
The patient received antibiotics therapy (2 grams of amoxicillin 500 mg 1 hour after surgery and Clavulin 785 mg 12/12 hours for 14 days). A previous oral bath with 0.12% chlorhexidine solution for 1 min is made. Local anesthesia (2% lidocaine with adrenaline 1: 100,000) was administered. In the first place, a supracrestal release incision was made with a total mucoperiosteal flap in order to expose the mandibular bone. Then, the element 46 was extracted and the alveolus effectively curetted. The mesial side of the implant 47 already had a significant bone loss, so we cured with a Teflon instruments from the Hu-Friedy brand, USA. Next, the line of separation of the bone was drawn with a Lindermam drill number 2 and the separation fracture made with ochibien chisels (Figure 3). The space between the fractures was filled with Blue Bone and covered by a HelitCote type 1 collagen membrane. Finally, the patient was instructed to avoid physical efforts for seven days and use 0.12% chlorhexidine gel, 3 times a day on the surgical wound for the period until removal of the stitches.
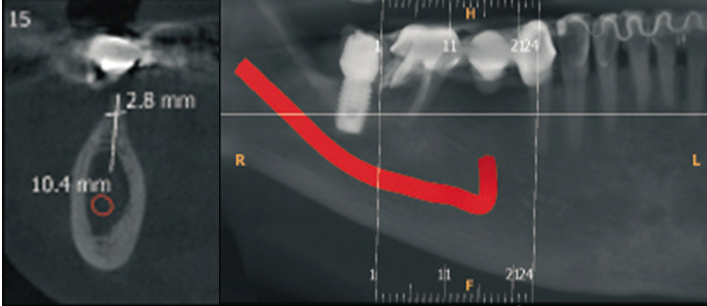
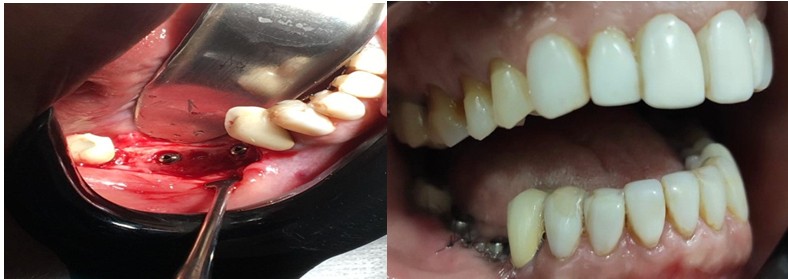
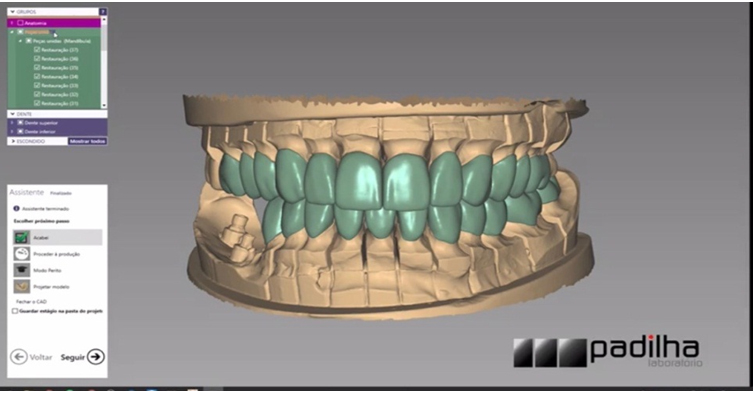
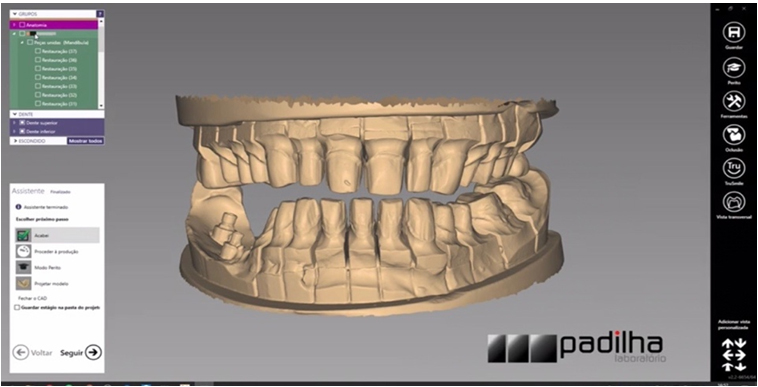
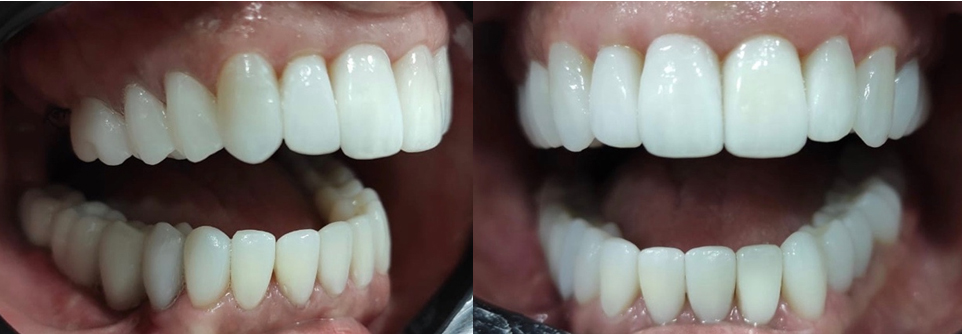
After split-crest surgery, the patient made periapical, panoramic radiography, and tomography. After waiting five months for the bone regeneration period, a bone gain was observed in the surgical area where it was performed by the 5 mm technique. For your information, the bone thickness at the site was of 2 mm, leading to a 3 mm bone gain, allowing the installation of 2 cone morse implants (Avant) in the grafted area in a surgical and prosthetic ideally position (Figure 4). Finally, after four months of the osseointegration period, the implants were activated and scanned to make the definitive ceramic prosthesis on implants and ceramic veneers in the remaining teeth (Figure 5, Figure 6, Figure 7, Figure 8, Figure 9, Figure 10, Figure 11, Figure 12).
Discussion
In a clinical study, presented a technique that combines the use of a piezoelectric scalpel and a conical bone expander, as a new way to perform split-crest procedures, in order to optimize the results and acceptability by the patients [13]. They conclude that all implants were successfully placed [14]. As the technique is very well accepted by patients, it has been progressing, and the new technologies improved their prognosis. Although the clinical study presented was made with a different technique, but the results are similar.
These authors made a study with 71 patients, in which 137 dental implants were installed in premolar or molar area. In order to monitor the long-term bone gain in split-crest technique, the bone increase procedure was performed in the determined areas and the implants followed a protocol to be installed in a second surgical part [15],[16]. Resulting in a vertical bone increase of an average of +0.89 ± 0.39 was achieved after almost 36 months after implant placement. This confirms the clinical case presented, which obtained a gain of 3 mm in thickness after the split-crest technique [17].
Starch-Jensen and Becktor. [18] in one comparative and four non-comparative studies met the inclusion criteria of this clinical case. Both treatment modalities revealed a high implant survival rate with few complications. High prosthesis survival rate and excellent implant stability values have been reported. In conclusion, the split-crest technique seems to be useful for the horizontal increase of alveolar defects on the maxilla with a high rate of prosthesis and implant survival. What is well described by other authors and corroborates the clinical case presented [3],[19],[20].
In another study it has been evaluated the survival rate of implants installed immediately after the split crest technique. Thirty-six patients were selected for this study. They underwent the placement of 93 dental implants in the toothless region after the split crest expansion procedure, with immediate loading procedure. The bone levels in the crest were measured at the beginning, in the temporary placement of the prosthesis, after one year and after two years with the definitive prothesis. A 98.92% survival rate has been reported with a 2-year follow-up [21],[22],[23]. In another study, six patients were evaluated by the split-crest technique using the immediate and late implantation after the split-crest surgery. As we have seen, they obtained results with no complication or loss of implants in both procedures with a follow-up of 27.8 months, indicating that this technique is a viable alternative for areas with lack of bone thickness [24], which corroborates with this case report and with other authors [25].
Conclusion
To summarize, the split-crest technique is very well based on the literature and can be indicated as a great alternative for cases of lack of bone thickness in the mandible; indicated with the use of biomaterial associated with collagen membrane.
REFERENCES
1.
Waechter J, Leite FR, Nascimento GG, Carmo Filho LC, Faot F. The split crest technique and dental implants: A systematic review and meta-analysis. Int J Oral Maxillofac Surg 2017;46(1):116–28. [CrossRef]
[Pubmed]

2.
Bassetti MA, Bassetti RG, Bosshardt DD. The alveolar ridge splitting/expansion technique: A systematic review. Clin Oral Implants Res 2016;27(3):310–24. [CrossRef]
[Pubmed]

3.
Garcez-Filho J, Tolentino L, Sukekava F, Seabra M, Cesar-Neto JB, Araújo MG. Long-term outcomes from implants installed by using split-crest technique in posterior maxillae: 10 years of follow-up. Clin Oral Implants Res 2015;26(3):326–31. [CrossRef]
[Pubmed]

4.
Barone A, Covani U. Maxillary alveolar ridge reconstruction with nonvascularized autogenous block bone: Clinical results. J Oral Maxillofac Surg 2007;65(10):2039–46. [CrossRef]
[Pubmed]

5.
Barone A, Varanini P, Orlando B, Tonelli P, Covani U. Deep-frozen allogeneic onlay bone grafts for reconstruction of atrophic maxillary alveolar ridges: A preliminary study. J Oral Maxillofac Surg 2009;67(6):1300–6. [CrossRef]
[Pubmed]

6.
da Silva Brum I, de Carvalho JJ, da Silva Pires JL, de Carvalho MAA, Dos Santos LBF, Elias CN. Nanosized hydroxyapatite and β-tricalcium phosphate composite: Physico-chemical, cytotoxicity, morphological properties and in vivo trial. Sci Rep 2019;9(1):19602. [CrossRef]

7.
Mestas G, Alarcón M, Chambrone L. Long-term survival rates of titanium implants placed in expanded alveolar ridges using split crest procedures: A systematic review. Int J Oral Maxillofac Implants 2016;31(3):591–9. [CrossRef]
[Pubmed]

8.
Brugnami F, Caiazzo A, Mehra P. Piezosurgery-assisted, flapless split crest surgery for implant site preparation. J Maxillofac Oral Surg 2014;13(1):67–72. [CrossRef]
[Pubmed]

9.
Simion M, Baldoni M, Zaffe D. Jawbone enlargement using immediate implant placement associated with a split-crest technique and guided tissue regeneration. Int J Periodontics Restorative Dent 1992;12(6):462–73.
[Pubmed]

10.
Hu GH, Froum SJ, Alodadi A, et al. A Three-stage split-crest technique: Case series of horizontal ridge augmentation in the atrophic posterior mandible. Int J Periodontics Restorative Dent 2018;38(4):565–73. [CrossRef]
[Pubmed]

11.
Cortese A, Pantaleo G, Amato M, Claudio PP. Ridge expansion by flapless split crest and immediate implant placement: Evolution of the technique. J Craniofac Surg 2016;27(2):e123–8. [CrossRef]
[Pubmed]

12.
Kelly A, Flanagan D. Ridge expansion and immediate placement with piezosurgery and screw expanders in atrophic maxillary sites: Two case reports. J Oral Implantol 2013;39(1):85–90. [CrossRef]
[Pubmed]

13.
Sammartino G, Cerone V, Gasparro R, Riccitiello F, Trosino O. The platform switching approach to optimize split crest technique. Case Rep Dent 2014;2014:850470. [CrossRef]
[Pubmed]

14.
Bruschi GB, Capparé P, Bravi F, et al. Radiographic evaluation of crestal bone level in split-crest and immediate implant placement: Minimum 5-year follow-up. Int J Oral Maxillofac Implants 2017;32(1):114–20. [CrossRef]
[Pubmed]

15.
Sanz-Sánchez I, Ortiz-Vigón A, Sanz-Martín I, Figuero E, Sanz M. Effectiveness of lateral bone augmentation on the alveolar crest dimension: A systematic review and meta-analysis. J Dent Res 2015;94(9 Suppl):128S–42S. [CrossRef]
[Pubmed]

16.
Berger S, Hakl P, Sutter W, et al. Interantral alveolar ridge splitting for maxillary horizontal expansion and simultaneous dental implant insertion: A case report. Ann Med Surg (Lond) 2019;48:83–7. [CrossRef]
[Pubmed]

17.
Waechter J, Leite FR, Nascimento GG, Carmo Filho LC, Faot F. The split crest technique and dental implants: A systematic review and meta-analysis. Int J Oral Maxillofac Surg 2017;46(1):116–28. [CrossRef]
[Pubmed]

18.
Starch-Jensen T, Becktor JP. Maxillary alveolar ridge expansion with split-crest technique compared with lateral ridge augmentation with autogenous bone block graft: A systematic review. J Oral Maxillofac Res 2019;10(4):e2. [CrossRef]
[Pubmed]

19.
Crespi R, Bruschi GB, Gastaldi G, Capparé P, Gherlone EF. Immediate loaded implants in split-crest procedure. Clin Implant Dent Relat Res 2015;17 Suppl 2:e692–8. [CrossRef]
[Pubmed]

20.
Arora V, Kumar D. Alveolar ridge split technique for implant placement. Med J Armed Forces India 2015;71(Suppl 2):S496–8. [CrossRef]
[Pubmed]

21.
Bassetti MA, Bassetti RG, Bosshardt DD. The alveolar ridge splitting/expansion technique: A systematic review. Clin Oral Implants Res 2016;27(3):310–24. [CrossRef]
[Pubmed]

22.
Shibuya Y, Yabase A, Ishida S, Kobayashi M, Komori T. Outcomes and treatments of mal fractures caused by the split-crest technique in the mandible. Kobe J Med Sci 2014;60(2):E37–42.
[Pubmed]

23.
Figliuzzi MM, Giudice A, Pileggi S, et al. Implant-prosthetic rehabilitation in bilateral agenesis of maxillary lateral incisors with a mini split crest. Case Rep Dent 2016;3591321. [CrossRef]
[Pubmed]

24.
Agabiti I, Botticelli D. Two-stage ridge split at narrow alveolar mandibular bone ridges. J Oral Maxillofac Surg 2017;75(10):2115.e1–2115.e12. [CrossRef]
[Pubmed]

25.
Sohn DS, Lee HJ, Heo JU, Moon JW, Park IS, Romanos GE. Immediate and delayed lateral ridge expansion technique in the atrophic posterior mandibular ridge. J Oral Maxillofac Surg 2010;68(9):2283–90. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Julio Marco Mainenti Rosalem - Conception of the work, Design of the work, Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Diogo Piacentini Engel - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Mateus Prates Bôa - Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Tatiana Nouira Y Maurity - Conception of the work, Design of the work, Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Igor da Silva Brum - Conception of the work, Design of the work, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2020 Julio Marco Mainenti Rosalem et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.