|
Case Report
Retroperitoneal ganglioneuroma in an adult
1 MBBS, School of Medicine, James Cook University, Townsville, Queensland 4814, Australia
2 MBBS FRACS, HPB & General Surgery Consultant, Department of General Surgery, Townsville University Hospital, Townsville, Queensland 4814, Australia
Address correspondence to:
Madeleine Louise Kelly
Louise Kelly, 1 James Cook Drive, Douglas, Townsville, Queensland 4814,
Australia
Message to Corresponding Author
Article ID: 100085Z12MK2021
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Kelly ML, Palamuthusingam P. Retroperitoneal ganglioneuroma in an adult. J Case Rep Images Surg 2021;7:100085Z12MK2021.ABSTRACT
Introduction: Retroperitoneal ganglioneuromas (GNs) are rare, benign neural tumors of the sympathetic nervous system
Case Report: A 57-year-old female presented with a two-week history of recurrent lower left sided back pain. Radiological investigations revealed a large paraaortic mass measuring 67 by 50 mm, extending from the adrenal gland to the mid lower pole of the left kidney, without lymphadenopathy elsewhere. Histopathological examination identified fragments composed of fibrous tissue, little adipose tissue with bland spindled cells and occasional ganglion cells, suggestive of ganglioneuroma. The decision was made for active surveillance over surgical excision.
Conclusion: Due to its rare nature and the nonspecific clinical signs, arriving at a definitive diagnosis remains a challenge for clinicians and no gold standard approach has been suggested.
Keywords: Adult, Diagnosis, Management, Retroperitoneal ganglioneuroma
Introduction
Accounting for 0.72–1.6% of primary retroperitoneal tumors, ganglioneuroma (GN) is a rare, benign tumor [1]. Typically, GN arises spontaneously in the sympathetic ganglion but can occur during radiation or chemotherapy and is commonly found in the posterior mediastinum or retroperitoneum [2]. Most retroperitoneal GNs are asymptomatic and are detected incidentally, due to the non-functionality of the tumor. When the tumor has metabolic activity caused by release of catecholamines, vasoactive intestinal polypeptides, or androgenic hormones, symptomatology of hypertension, diarrhea, and virilization may be seen [3],[4]. Owing to the size of the tumor several complications can occur due to compression of adjacent organs [4].
Case Report
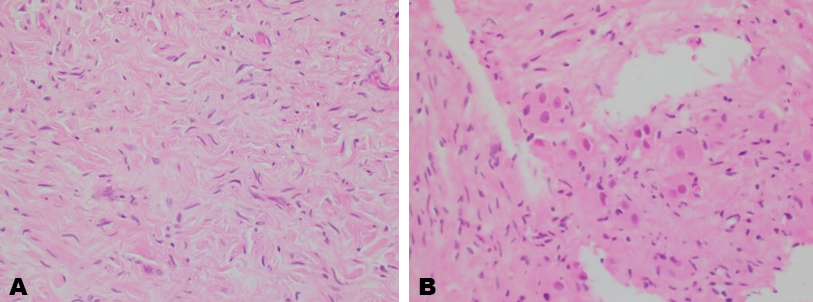
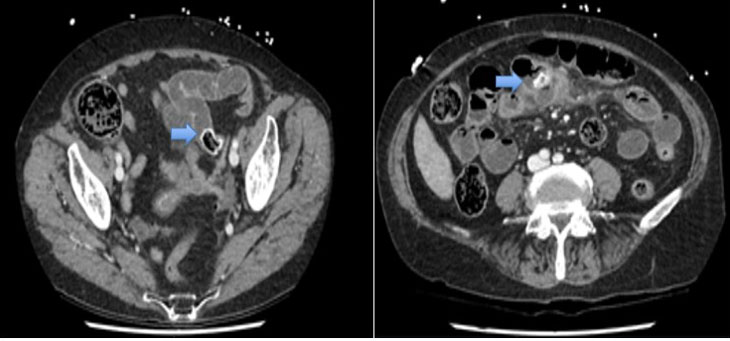
A 57-year-old female presented with a two-week history of left sided lower back pain associated with night sweats. No other constitutional symptoms were reported, and the patient’s physical examination was unremarkable. Past medical history of note included ischemic heart disease managed medically. The patient’s blood panel was within normal ranges, of note, serum electrolyte and hormonal studies (including CgA and plasma metanephrines) were normal, confirming the non-functional nature of the mass. Computed tomography (CT) revealed a large paraaortic mass, measuring 67 by 50 mm, extending from the adrenal gland to the mid lower pole of the left kidney (Figure 1A and Figure 1B). The mass enveloped the coeliac axis and superior mesenteric artery (SMA). No lymphadenopathy elsewhere was shown on positron emission tomography (PET) scan and fluorodeoxyglucose avidity was relatively low at 3.36, suggestive of no metabolic activity. Computed tomography guided biopsy of the mass was performed with the specimen sent for histopathological examination. This identified fragments composed of fibrous tissue and little adipose tissue with bland spindled cells (Figure 2A) and occasional ganglion cells (Figure 2B). No atypia or mitotic activity was evident. Flow cytometry was inconclusive for surface marker analysis. Histopathological examination was consistent with a diagnosis of ganglioneuroma. Due to the extensive vascular involvement of the coeliac axis and superior mesenteric artery, surgical excision was not considered to be appropriate and instead, due to the benign nature of the tumor and the mild symptomatology of back pain, regular surveillance was preferred. The plan for follow-up was for annual CT bought forward if symptomatology worsened, with re-evaluation of the surgical plan if required. Since diagnosis the patient has experienced an episode of back pain every six months, which resolves within a week. Further radiological imaging showed a stable lesion, with minimal growth, with a maximal transverse diameter of 81 mm and maximal height of 94.5 mm (Figure 1C). This case highlights a rare cause of back pain and retroperitoneal mass in an adult, and the diagnostic challenge that arises with this rare tumor.
Discussion
Retroperitoneal ganglioneuromas (GNs) are a rare, benign tumor of neural crest origin, commonly arising from anywhere along the sympathetic plexus, and occasionally in the adrenal medulla [1],[2]. It is commonly found in the posterior mediastinum (41.5%) or the retroperitoneum (37.5%) [3],[4],[5]. Typically, GN arises spontaneously in the sympathetic ganglion but can occur during radiation or chemotherapy. Traditionally affecting females more than males, GN tumors are composed of mature Schwann cells, ganglion cells, and nerve fibers [1],[6]. Mostly of sporadic origin, some familial predisposition has been documented and there is an association with Turners syndrome and multiple endocrine neoplasia II [7]. Most GN tumors are asymptomatic. Rarely the tumor will be diagnosed due to its secretory activity of catecholamines, vasoactive intestinal polypeptides, or androgenic hormones, producing symptomatology of hypertension, diarrhea, and virilization. The compressive effects of the tumor on neighboring tissues can also cause complications. The slow growth pattern of GN tumors results in diagnosis more commonly in older children and young adults [1],[6].
Given its asymptomatic nature and lack of specific diagnostic methods, imageology plays an important role in this diagnosing disease. Computed tomography, ultrasound, and magnetic resonance imaging show a well-circumscribed, oval, lobulated mass. Ultrasound reveals a homogenous, hypoechoic, mass while an unenhanced CT will show a low attenuation, homogenous mass. Calcifications are seen in about 20% of cases [6]. Ganglioneuromas tend to surround or displace major blood vessels but do not cause occlusion or compression [1]. There are no specific radiological findings for GN tumors making differentiation from other tumors difficult and often only possible with histological examination of the resected mass postoperatively. Cellular atypia, mitotic activity, and necrosis are not observed on histopathological examination [8]. Definitive diagnosis is confirmed after pathological examination of the specimen.
Gold standard management remains to be determined with questions arising as to whether surveillance or surgical excision of an asymptomatic, benign lesion is preferred. With most tumors discovered incidentally while investigating nonspecific symptoms, currently there are no surveillance guidelines for retroperitoneal GN [6],[9]. Reports have suggested that for tumors less than 6 cm in size with no metabolic activity or for tumors not amenable to complete resection that conservative follow-up is an appropriate option but time intervals vary, guided by patient symptoms [10]. Other reports suggest due to the difficult preoperative diagnostic nature of this lesion, that surgical resection is best especially in cases of marked growth in tumor size, increased secretory activity, or encroachment on vertebral foramina [1],[9],[10]. Potential alternative interventions include fractionated resection of GN that surround major vessels and transarterial embolization using both microparticles and a liquid embolic agent, a technique that may be a safe and effective option, particularly for symptomatic relief [11]. In general, surgery is the only potential curative treatment, with a low reoccurrence rate after tumor resection.
Conclusion
Retroperitoneal ganglioneuromas are well differentiated benign tumors that are typically hormonally inactive. Although an uncommon lesion in adults, it should be considered among potential diagnosis for any retroperitoneal mass. Due to its rare nature, radiological and pathological investigations provide important diagnostic information for effective management; however, the gold standard management is yet to be determined. Guidelines on surveillance versus surgical resection remain dependent on patient preference and the symptomatic effect on patient quality of life. Retroperitoneal ganglioneuromas removed surgically have an excellent prognostic outcome.
REFERENCES
1.
Geoerger B, Hero B, Harms D, Grebe J, Scheidhauer K, Berthold F. Metabolic activity and clinical features of primary ganglioneuromas. Cancer 2001;91(10):1905–13. [CrossRef]
[Pubmed]

3.
Rha SE, Byun JY, Jung SE, Chun HJ, Lee HG, Lee MJ. Neurogenic tumors in the abdomen: Tumor types and imaging characteristics. Radiographics 2003;23(1):29–43. [CrossRef]
[Pubmed]

4.
Radin R, David CL, Goldfarb H, Francis IR. Adrenal and extra-adrenal retroperitoneal ganglioneuroma: Imaging findings in 13 adults. Radiology 1997;202(3):703–7. [CrossRef]
[Pubmed]

5.
Lonergan GJ, Schwab CM, Suarez ES, Carlson CL. Neuroblastoma, ganglioneuroblastoma, and ganglioneuroma: Radiologic-pathologic correlation. Radiographics 2002;22(4):911–34. [CrossRef]
[Pubmed]

6.
Ichikawa T, Ohtomo K, Araki T, et al. Ganglioneuroma: Computed tomography and magnetic resonance features. Br J Radiol 1996;69(818):114–21. [CrossRef]
[Pubmed]

7.
Moriwaki Y, Miyake M, Yamamoto T, et al. Retroperitoneal ganglioneuroma: A case report and review of the Japanese literature. Intern Med 1992;31(1):82–5. [CrossRef]
[Pubmed]

8.
Joshi VV. Peripheral neuroblastic tumors: Pathologic classification based on recommendations of international neuroblastoma pathology committee (modification of Shimada classification). Pediatr Dev Pathol 2000;3(2):184–99. [CrossRef]
[Pubmed]

9.
Joshi VV. Peripheral neuroblastic tumors: Pathologic classification based on recommendations of international neuroblastoma pathology committee (modification of shimada classification). Pediatr Dev Pathol 2000;3(2):184–99. [CrossRef]
[Pubmed]

10.
Bove KE, McAdams AJ. Composite ganglioneuroblastoma. An assessment of the significance of histological maturation in neuroblastoma diagnosed beyond infancy. Arch Pathol Lab Med 1981;105(6):325–30.
[Pubmed]

11.
Burke C, Salsamendi J, Bhatia SS, Pitcher JD Jr. Transarterial embolization of large retroperitoneal ganglioneuromas. J Vasc Interv Radiol 2014;25(3):490–3. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Madeleine Louise Kelly - Conception of the work, Design of the work, Acquisition of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Pranavan Palamuthusingam - Conception of the work, Design of the work, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
AcknowledgmentsSpecial thanks to Dr. Peter Kanowski for providing pathology images for this paper.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2021 Madeleine Louise Kelly et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.