|
Case Report
False-positive findings on 18-Fluoro-deoxy-glucose positron emission tomography due to granulomatous inflammation in a rectal cancer patient with an extensive tattoo
1 Department of Visceral, Transplant and Thoracic Surgery, Center of Operative Medicine, Medical University of Innsbruck, Innsbruck, Austria
2 Department of Radiology, Medical University of Innsbruck, Innsbruck, Austria
Address correspondence to:
Reinhold Kafka-Ritsch
Department of Visceral, Transplant and Thoracic Surgery, Center of Operative Medicine, Medical University of Innsbruck, Anichstraße 35, 6020 Innsbruck,
Austria
Message to Corresponding Author
Article ID: 100102Z12KE2022
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Esswein K, Gassner E, Bale R, Kafka-Ritsch R. False-positive findings on 18-Fluoro-deoxy-glucose positron emission tomography due to granulomatous inflammation in a rectal cancer patient with an extensive tattoo. J Case Rep Images Surg 2022;8(1):16–21.ABSTRACT
Introduction: In patients with colorectal cancer a combined 18-Fluoro-deoxy-glucose positron emission tomography with computed tomography (CT) may be useful in advanced or recurrent disease to assess distant metastases. In the interpretation several pitfalls must be considered.
Case Report: A 37-year-old woman presented with metachronous liver metastases three years after the first diagnosis of rectal cancer. A second-line chemotherapy and stereotactic radiofrequency ablation was performed. Six months after the intervention, the follow-up magnetic resonance imaging revealed new liver lesions with indefinite diffusion impairment and pathologic contrast enhancement in portal venous and hepatobiliary phase. Further investigation with combined 18-Fluoro-deoxy-glucose positron emission tomography with CT showed for malignancy suspicious glucose-uptake in the respective liver lesions and additional in unilateral enlarged inguinal lymph nodes. Because of the uncommon findings, histologic specimen was obtained. Histology revealed no malignancy, but granulomatous hepatitis and lymphadenitis consistent with sarcoid-like reaction. Furthermore, deposition of tattoo pigment was found in the enlarged lymph nodes, most likely deriving from a large tattoo located on the right lower extremity.
Conclusion: We describe a case of a rectal cancer patient with misleading imaging findings on combined 18-Fluoro-deoxy-glucose positron emission tomography with CT. Awareness of rare conditions such as granulomatous inflammation and false-positive findings is key in the presence of uncommon findings, like in this case, suggested spread outside the metastatic pathway.
Keywords: Colorectal liver metastases, Magnetic resonance imaging, Sarcoid-like reaction, Sarcoidosis, Stereotactic radiofrequency ablation
Introduction
Colorectal cancer is the third most common malignant neoplasms and the second leading cause for cancer-related death worldwide [1]. The most frequent sites of metastatic colorectal cancer disease are liver followed by lung. Metachronous liver metastases develop in about 5–15% of the patients [2],[3],[4]. Even in metastatic tumor disease, a curative treatment approach can be feasible. Besides a surgical resection of colorectal liver metastases (CRLM), interventional radiological procedures such as thermal ablation become more important.
The European Conference on Interventional Oncology and European Society of Oncologic Imaging (ECIO-ESOI) consensus guidelines 2018 recommend both, magnetic resonance imaging (MRI) and combined 18-Fluoro-deoxy-glucose positron emission tomography with CT (18-FDG-PET/CT) for follow-up after thermal ablation of CRLM, although comparative literature is sparse [5]. A slight preference for MRI as primary modality is given in the recommended protocol owing to superior spatial resolution. For follow-up of patients after thermal ablation of CRLM, 18-FDG-PET/CT is suggested as a problem solver, additionally, it is useful to assess distant metastases. Several pitfalls must be considered in the interpretation. Next to false-positive findings by infection, inflammatory response to tattoo pigment mimicking a positive lymph node was reported [6],[7],[8]. We present a case in which not only inflammatory response to tattoo pigment but also granulomatous inflammation adjacent to a coagulative necrosis after liver thermal ablation led to false-positive findings.
Case Report
The patient is a 37-year-old woman with rectal cancer and an initial tumor classification of pT3N1M0. A preoperative chemoradiotherapy, oncological rectal resection followed by an adjuvant chemotherapy with oxaliplatin and capecitabine (XELOX) was performed. Three years after the first diagnosis, metachronous liver metastases were detected in segment IVa/VIII and segment VIII. Therefore, in a curative attempt, the patient was assigned to a second-line chemotherapy with folinic acid, fluorouracil, irinotecan (FOLFIRI), and panitumumab as well as subsequent liver stereotactic radiofrequency ablation (SRFA). Six months after SRFA, a follow-up magnetic resonance imaging (MRI) gave the suspicion of recurring liver metastases adjacent to the coagulative necrosis.
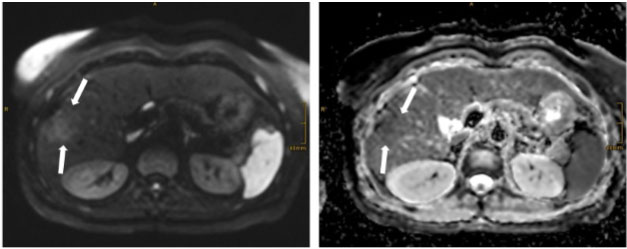
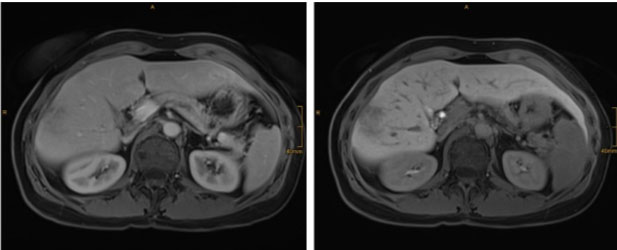
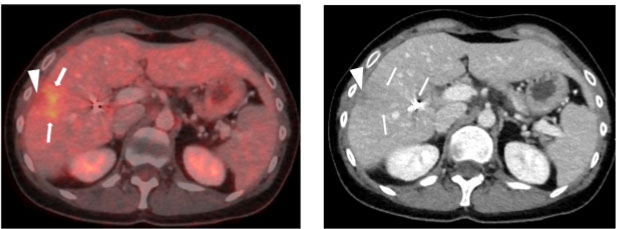
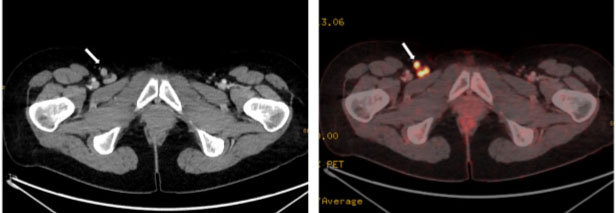
The follow-up imaging was performed with MRI using the following sequences: T1 in/opposed phase, T2 with and without fat saturation, T1 3D sequences pre-contrast and contrast dynamics in arterial, portal-venous, delayed, and hepatobiliary phase, as well as diffusion-weighted images (DWIs). A small area with elevated signal on diffusion-weighted images at b=1000 s/mm2 was found adjacent to the coagulative necrosis. Corresponding apparent diffusion coefficient (ADC) maps yielded subtle signal loss, indicative of mild cell barrier pathology (Figure 1). Contrast-enhanced MRI during portal venous phase and especially during hepatobiliary (HBP) phase showed ill-defined hypointensities (Figure 2). Diffusion-weighted images and hepatobiliary phase images generated the suspicion of recurrence and prompted an 18-FDG-PET/CT as a second imaging modality. 18-Fluoro-deoxy-glucose positron emission tomography with CT scans revealed an indistinct 18-FDG-avid focus next to the ablation zone, with corresponding contrast-enhanced CT showing no definite changes (Figure 3). Additionally, the 18-FDG-PET/CT revealed enlarged inguinal lymph nodes on the right side, which were 18-Fluoro-deoxy-glucose avid, too (Figure 4).
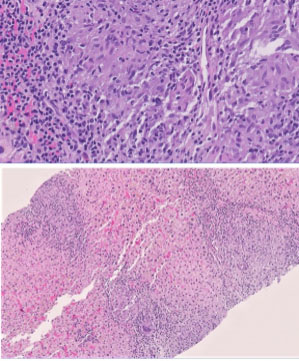
These diagnostic findings were suspicious for systemic disease and potentially directed the patient toward palliative treatment. Considering the unusual lymph node localization for T3 rectal cancer, ambiguous findings on MRI and the patient’s young age, histologic specimen of the lymph nodes, and the newly detected liver lesions were obtained through inguinal lymphadenectomy and percutaneous liver biopsy, respectively. During lymphadenectomy, intraoperatively a black coloring of the lymph nodes was noticed. In histology a deposition of black pigment consistent with tattoo ink was found, which was most likely related to a large unilateral tattoo on the right leg (Figure 5). Furthermore, epithelioid granuloma was found in both the liver biopsies and right inguinal lymph nodes consistent with a sarcoid-like reaction, but no malignant cells (Figure 6). Special staining excluded infectious granuloma caused by mycobacteria or fungi. In addition, despite granulomatous inflammation in histologic specimen, the patient showed no cutaneous, pulmonary, or systemic symptoms nor laboratory evidence of systemic sarcoidosis. Therefore, no specific therapy was started.
As the histologic examination of the inguinal lymph nodes and liver biopsy showed no malignant cells, cancer specific therapy was put on hold and the patient was included in a colorectal cancer surveillance protocol. One year later the patient is still cancer free.
Discussion
This case illustrates false-positive findings on 18-FDG-PET/CT and MRI caused by inflammatory nodal response to tattoo pigment with sarcoid-like granuloma formation as well as granulomatous hepatitis adjacent to a coagulative necrosis after liver SRFA. The finding of false-positive lymph nodes in 18-FDG-PET/CT caused by inflammatory response to tattoo pigment has been reported in several cases of cancer patients [6],[7],[8]. Excess tattoo pigments are transported to regional lymph nodes and can result in foreign body reaction with pigment uptake by macrophage and granuloma formation even years after the initial tattoo was made [9],[10]. Awareness for this pitfall in the interpretation is important as the number of people with tattoos is rising and thus the rate of false-positive results is expected to increase. The presence and location of a tattoo should be documented as a clinical information and considered in the interpretation of an 18-FDG-PET/CT. In our case, detection of suspect unilateral lymph nodes in the drainage pathway of a big tattoo should have raised doubts.
While reactive granulomatous lesions usually are restricted to the tattooed skin area and drainage basin, a coincidence of systemic sarcoidosis and tattoo has been described [11],[12],[13]. Yet, the correlation and distinction between local tattoo reactions and systemic sarcoidosis is controversial [14]. In our case, in addition to granulomatous lymphadenitis, sarcoid-like hepatitis was also found in biopsies obtained from areas bordering an SRFA-induced necrosis. No pigment deposition was described in specimen from the liver. The co-occurrence of malignancy and either regional lympho-nodal or systemic sarcoid-like reaction is recognized. This includes also multisystem sarcoid-like reactions to chemotherapy-like oxaliplatin, also administered to our patient [15]. In a study published in 2009 by Chowdhury et al. sarcoid-like reactions were more often detected in restaging than in primary staging PET/CT, similar to our case [16]. Although, the underlying mechanism how malignancy is leading to granulomatous reaction is not fully elucidated [17]. In our case the SRFA itself may have induced the sarcoid-like reaction as the suspicious lesions were adjacent to the coagulative necrosis after liver SRFA. This is a new information, unless we could not find publications of a similar case.
Sarcoid-like granulomas cause high levels of 18-FDG uptake, similar to systemic sarcoidosis during phases of inflammatory activity [16],[18]. In cancer patients with sarcoid-like granulomas, 18-FDG-PET/CT therefore cannot differentiate between malignancy and inflammatory granulomatous reaction. To the best of our knowledge, there is no available evidence on MRI features of sarcoid-like granulomas of the liver. A case report could demonstrate a splenic manifestation in a colorectal cancer patient with a hypodense mass on CT [19]. Few reports focus on hepatosplenic MR appearance of idiopathic multisystem sarcoidosis and the majority of cases present with diffuse hepatosplenomegaly due to non-discernible micronodules. In the rare forms of hepatosplenic sarcoidosis with large nodules (5–15 mm), restricted MR diffusion is correlated to disease activity. On gadolinium-enhanced T1 sequences in the portal-venous phase, sarcoid macrogranuloma should appear hypointense [20],[21]. In retrospect, both DWI intensity and hypointense alteration on portal venous phase images are, although non-specific, consistent with granulomatous liver disease, a diagnosis not taken into account due to its absolute rarity.
Diffusion-weighted MRI offers functional tissue assessment with restricted diffusion as a marker for cellularity and pathologic cell barriers [22]. On DWI at high b-values (b=800 to 1000 s/mm2), diffusion-restricted areas remain signal-intense, thus bright. Calculating an apparent diffusion coefficient (ADC value) in multiples of 10-3 mm2/s from DWI allows for quantification of restricted diffusion. On ADC maps, low values, thus dark areas, represent restriction. The role of DWI for characterizing periablative zones remains unclear. Tissue damage surrounding thermal necrosis may result in restricted diffusion due to cellular membrane breakdown and perfusion inhomogenities. Published evidence suggests that periablative diffusion-restriction during the first 200 days after RFA is benign in the majority of cases [23]. Secondly, ADC values in malignant diffusion-restriction tend to be lower compared to benign DWI changes, but there is an overlap. In our case, ADC values were marginally lower than normal liver, favoring the diagnosis of malignant tissue ingrowth. Magnetic resonance signal intensity during hepatobiliary phase of gadoxetic acid enhanced MRI correlates to the presence of functioning hepatocytes with expression of specific membrane transporters [24]. Consequently, hypointense changes during HBP are due to non-functional hepatocytes, such as in classical adenoma, or due to replacement by histopathologically different cells, such as in metastases [25],[26]. Hypointensity on HBP images in the presented patient was hence suggestive of infiltration by recurrent metastatic tissue.
Imaging after RFA follows a sequential pattern of periablation changes representing early edema, followed by inflammatory hyperemia with resolution after 4–9 months [27]. Accordingly, CT and MRI during the first 24–48 hours reflect edema with a hypodense rim on CT, reduced signal on T1-weighted MRIs and elevated T2 signal. Subsequently, periablative inflammation is represented by increased contrast-enhancement during arterial dominant phases on CT and MRI, as a marker of vascularity. Of note, imaging correlates of inflammation are rather homogeneous than nodular. On 18-FDG-PET/CT, a peripheral uniform rim of 18-FDG uptake as a surrogate marker for inflammation is seen from day 2 after ablation and gradually decreases during the first six months after RFA, when inflammatory changes have resolved. In our patient, while 18-FDG uptake at six months after RFA might represent prolonged inflammation, MRI diffusion-restriction and more specifically, hypointense changes on hepatobiliary MRIs raised the suspicion of periablative recurrence.
Conclusion
This case demonstrates that various distinct processes (e.g., tattoo pigment, chemotherapeutic drugs, malignancy) may lead to granulomatous inflammation which must not be diagnosed false-positively in 18-FDG-PET/CT as metastatic disease causing false therapeutic decisions. Therefore, cautious interpretation of follow-up diagnostics in such patients is crucial and in patients with non-specific findings or untypical location a histological assessment should be performed before determining the definitive treatment algorithm.
REFERENCES
1.
Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021;71(3):209–49. [CrossRef]
[Pubmed]

2.
Engstrand J, Nilsson H, Strömberg C, Jonas E, Freedman J. Colorectal cancer liver metastases – A population-based study on incidence, management and survival. BMC Cancer 2018;18(1):78. [CrossRef]
[Pubmed]

3.
Manfredi S, Lepage C, Hatem C, Coatmeur O, Faivre J, Bouvier AM. Epidemiology and management of liver metastases from colorectal cancer. Ann Surg 2006;244(2):254–9. [CrossRef]
[Pubmed]

4.
Hackl C, Neumann P, Gerken M, Loss M, Klinkhammer-Schalke M, Schlitt HJ. Treatment of colorectal liver metastases in Germany: A ten-year population-based analysis of 5772 cases of primary colorectal adenocarcinoma. BMC Cancer 2014;14:810. [CrossRef]
[Pubmed]

5.
Maas M, Beets-Tan R, Gaubert JY, et al. Follow-up after radiological intervention in oncology: ECIO-ESOI evidence and consensus-based recommendations for clinical practice. Insights Imaging 2020;11(1):83. [CrossRef]
[Pubmed]

6.
Marom EM, Ofek E, Bekker E, Onn A. Tattoo-induced false-positive FDG PET/CT interpretation while staging for lung cancer. J Thorac Oncol 2018;13(4):585–6. [CrossRef]
[Pubmed]

7.
Nam H, Smith S, Laing R. A pitfall of 18-fluorodeoxyglucose-PET in a patient with a tattoo. Lancet Oncol 2007;8(12):1147–8. [CrossRef]
[Pubmed]

8.
Grove N, Zheng M, Bristow RE, Eskander RN. Extensive tattoos mimicking lymphatic metastasis on positron emission tomography scan in a patient with cervical cancer. Obstet Gynecol 2015;126(1):182–5.
[Pubmed]

9.
Serup J, Carlsen KH, Sepehri M. Tattoo complaints and complications: Diagnosis and clinical spectrum. Curr Probl Dermatol 2015;48:48–60. [CrossRef]
[Pubmed]

10.
Bäumler W. Absorption, distribution, metabolism and excretion of tattoo colorants and ingredients in mouse and man: The known and the unknown. Curr Probl Dermatol 2015;48:176–84. [CrossRef]
[Pubmed]

11.
Morales-Callaghan AM Jr, Aguilar-Bernier M Jr, Martínez-García G, Miranda-Romero A. Sarcoid granuloma on black tattoo. J Am Acad Dermatol 2006;55(5 Suppl):S71–3. [CrossRef]
[Pubmed]

12.
Shinohara MM, Nguyen J, Gardner J, Rosenbach M, Elenitsas R. The histopathologic spectrum of decorative tattoo complications. J Cutan Pathol 2012;39(12):1110–8. [CrossRef]
[Pubmed]

13.
Simunovic C, Shinohara MM. Complications of decorative tattoos: Recognition and management. Am J Clin Dermatol 2014;15(6):525–36. [CrossRef]
[Pubmed]

14.
Bose R, Sibley C, Fahim S. Granulomatous and systemic inflammatory reactions from tattoo ink: Case report and concise review. SAGE Open Med Case Rep 2020;8:2050313X20936036. [CrossRef]
[Pubmed]

15.
Aedma SK, Chidharla A, Kelting S, Kasi A. Oxaliplatin-associated sarcoid-like reaction masquerading as recurrent colon cancer. BMJ Case Rep 2020;13(9):e229548. [CrossRef]
[Pubmed]

16.
Chowdhury FU, Sheerin F, Bradley KM, Gleeson FV. Sarcoid-like reaction to malignancy on whole-body integrated (18)F-FDG PET/CT: Prevalence and disease pattern. Clin Radiol 2009;64(7):675–81. [CrossRef]
[Pubmed]

17.
Cohen PR, Kurzrock R. Sarcoidosis and malignancy. Clin Dermatol 2007;25(3):326–33. [CrossRef]
[Pubmed]

18.
Akaike G, Itani M, Shah H, et al. PET/CT in the diagnosis and workup of sarcoidosis: Focus on atypical manifestations. Radiographics 2018;38(5):1536–49. [CrossRef]
[Pubmed]

19.
Shima T, Tanaka Y, Katsuragi K, et al. Sarcoid reaction in the spleen after sigmoid colon cancer resection: A case report. Surg Case Rep 2016;2(1):115. [CrossRef]
[Pubmed]

20.
Palmucci S, Torrisi SE, Caltabiano DC, et al. Clinical and radiological features of extra-pulmonary sarcoidosis: A pictorial essay. Insights Imaging 2016;7(4):571–87. [CrossRef]
[Pubmed]

21.
Masuda K, Takenaga S, Morikawa K, Kano A, Ojiri H. Hepatic sarcoidosis with atypical radiological manifestations: A case report. Radiol Case Rep 2018;13(5):936–9. [CrossRef]
[Pubmed]

22.
Tang L, Zhou XJ. Diffusion MRI of cancer: From low to high b-values. J Magn Reson Imaging 2019;49(1):23–40. [CrossRef]
[Pubmed]

23.
Schraml C, Schwenzer NF, Clasen S, et al. Navigator respiratory-triggered diffusion-weighted imaging in the follow-up after hepatic radiofrequency ablation-initial results. J Magn Reson Imaging 2009;29(6):1308–16. [CrossRef]
[Pubmed]

24.
Kitao A, Zen Y, Matsui O, et al. Hepatocellular carcinoma: Signal intensity at gadoxetic acid-enhanced MR Imaging—correlation with molecular transporters and histopathologic features. Radiology 2010;256(3):817–26. [CrossRef]
[Pubmed]

25.
Bhayana R, Baliyan V, Kordbacheh H, Kambadakone A. Hepatobiliary phase enhancement of liver metastases on gadoxetic acid MRI: Assessment of frequency and patterns. Eur Radiol 2021;31(3):1359–66. [CrossRef]
[Pubmed]

26.
Fujita N, Nishie A, Asayama Y, et al. Hyperintense liver masses at hepatobiliary phase gadoxetic acid-enhanced MRI: Imaging appearances and clinical importance. Radiographics 2020;40(1):72–94. [CrossRef]
[Pubmed]

27.
Sainani NI, Gervais DA, Mueller PR, Arellano RS. Imaging after percutaneous radiofrequency ablation of hepatic tumors: Part 1, Normal findings. AJR Am J Roentgenol 2013;200(1):184–93. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Katharina Esswein - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Eva Gassner - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Reto Bale - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Reinhold Kafka-Ritsch - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2022 Katharina Esswein et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.