|
Case Report
Life-threatening upper gastrointestinal hemorrhage from a Cameron ulcer
1 Surgical Trainee, Tamworth Rural Referral Hospital, Tamworth, NSW, Australia
2 Consultant General Surgeon, Tamworth Rural Referral Hospital, Tamworth, NSW, Australia
Address correspondence to:
Alen Brodaric
Royal Prince Alfred Hospital, 50 Missenden Road, Camperdown, NSW 2050,
Australia
Message to Corresponding Author
Article ID: 100128Z12AB2024
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Brodaric A, Srinivasan R. Life-threatening upper gastrointestinal hemorrhage from a Cameron ulcer. J Case Rep Images Surg 2024;10(1):1–5.ABSTRACT
Introduction: Acute upper gastrointestinal bleeding can be life-threatening; however, Cameron lesions are an uncommon etiology. These lesions should be suspected with a previous history or endoscopic evidence of hiatus hernia.
Case Report: A male in his 60s presented to the emergency department with hematemesis and melena for two days. He had no prior medical, surgical, or endoscopic history and took no regular medications. He was resuscitated with packed red blood cell transfusion and treated with proton pump inhibitor infusion then transferred to operating theaters for endoscopic hemostasis. Cardiac arrest occurred after anesthetic induction and airway intubation, return of spontaneous circulation was achieved after cardiopulmonary resuscitation and further blood transfusion. At endoscopy a Cameron ulcer was noted in the hiatus hernia with overlying blood clot with no active hemorrhage, treated with Hemospray®.
Conclusion: Patients presenting with acute upper gastrointestinal hemorrhage and shock need urgent upper gastrointestinal (UGI) endoscopy. Topical hemostatic agents are successful in controlling bleeding when challenging anatomy precludes use of banding or clip application.
Keywords: Cameron lesion, Emergency endoscopy, Upper gastrointestinal hemorrhage
Introduction
Upper gastrointestinal (UGI) bleeding has multiple etiologies; however, massive bleeding commonly requires a degree of portal hypertension or ulcer erosion through an artery. Cameron lesions are caused by diaphragmatic erosion of gastric mucosa in patients with a hiatus hernia. Cameron lesions may cause an insidious iron deficiency anemia due to chronic low volume gastrointestinal bleeding but rarely massive hemorrhage. We present an unusual case of massive bleeding causing hemorrhagic shock and cardiac arrest from a Cameron lesion.
Case Report
A male in his 60s presented to our emergency department via ambulance with a two-day history of hematemesis and melena. He had sudden onset of symptoms with no precipitating factors including forceful vomiting and he had no previous medical, surgical, or medication history. He denied having previous endoscopies. He had no risk factors for cirrhosis and no family history of peptic ulcer disease. He did not drink alcohol or smoke tobacco. On arrival, he was tachycardic to 100 beats per minute, blood pressure was 124/65 mmHg. Oxygen saturation was 98% on room air and respiratory rate was 16 breaths per minute. On examination, his skin and clothing had fresh blood stains. He was pale and not icteric. His abdomen was soft with no discernible tenderness or organomegaly. Per rectal examination yielded melena without any rectal masses.
Paramedics reported hematemesis and tachycardia during hospital transfer and he received 1 L of sodium chloride 0.9% resuscitation. In the emergency department, he had ongoing hematemesis prompting activation of massive transfusion protocol due to ongoing hemorrhage, hemodynamic instability (HR 125, BP 118/84), and anticipated transfusion of >50% total blood volume in 4 hours. Furthermore, he was treated with high dose proton pump inhibitor infusion and informed consent was given for upper gastrointestinal endoscopy, with a view to proceed for laparotomy if endoscopic hemostasis failed.
His hemoglobin count was 84 g/L (130–180 g/L), platelet count was 90 × 109/L (150–400 × 109/L), and white cell count was 19.0 × 109/L (4.0–11.0 × 109/L).
His urea was 22.0 mmol/L (4.0–9.0 mmol/L), creatinine 116 µmol/L (60–110 umol/L), and estimated glomerular filtration rate (eGFR) 55 ml/min/1.73 m2 (>60 mL/min/1.73 m2). His electrolytes were normal, sodium 141 mmol/L (135–145 mmol/L), potassium 4.0 mmol/L (3.5–5.2 mmol/L), chloride 110 mmol/L (95–110 mmol/L) with an anion gap of 18 mmol/L (7–17 mmol/L).
Liver function tests showed bilirubin 20 (<20 µmol), albumin 35 (30–44 g/L), protein 59 (60–80 g/L), gamma-glutamyl transferase 58 (5–50 U/L), alkaline phosphatase 70 (30–110 U/L), alanine transaminase 14 (10–50 U/L), and aspartate aminotransferase 19 (10–35 U/L).
His prothrombin time was 13 seconds (9–13 seconds), activated partial thromboplastic time was less than 22 seconds (25–37 seconds), and fibrinogen was 1.9 g/L (2.0–4.6 g/L).
Venous blood gas showed a pH of 7.38 (7.30–7.40), carbon dioxide of 33 mmHg (40–50 mmHg), bicarbonate of 20 mmol/L (22–32 mmol/L), lactate of 5.9 mmol/L (<2 mmol/L), and base excess of -5.2 mmol/L (-3.0 to 3.0 mmol/L).
Differential diagnosis
The differential diagnosis for upper gastrointestinal bleeding in this patient includes:
- Peptic ulcer disease (Helicobacter pylori infection, gastrinoma, Zollinger–Ellison syndrome).
- Variceal bleeding from portal hypertension (cirrhosis, portal vein thrombosis, and idiopathic) though he had no risk factors.
- Occult gastric or duodenal neoplasia.
- Crohn’s disease of the stomach/duodenum with ulceration.
- Gastric antral vascular ectasia (GAVE).
- Mallory–Weiss tear.
- Dieulafoy lesion.
- Aneurysm involving the gastroduodenal artery.
This patient had acute onset of disease without known risk factors. Large volume bleeding would generally imply severe disease of any manifestation. In this case upper gastrointestinal endoscopy was urgently needed for hemorrhage control and diagnosis.
Pre-operative course
The patient’s heart rate normalized, and blood pressure remained stable after administration of proton pump inhibitor and two units of packed red cells. An arterial line was inserted. He remained stable during rapid sequence induction (Propofol 100 mg, Fentanyl 100 mcg, and Rocuronium 50 mcg) and endotracheal intubation (due to aspiration risk) was successful. After intubation, the last recorded non-invasive blood pressure was 110/54, oxygen saturation 100% on FiO2 100%; however, the arterial line had no discernible waveform. Given the patient was sedated with muscle relaxant onboard, clinical diagnosis of cardiac arrest was made due to the absence of carotid and femoral pulses. Cardiopulmonary resuscitation (CPR) was commenced by the operative team leading to oral regurgitation of frank blood around the endotracheal tube. During two rounds of 2 minutes CPR, there was pulseless electrical activity on rhythm assessment, 1 gram of intravenous adrenaline, and 75 milligrams of amiodarone were administered. Transfusion of two further units of packed red blood cells prompted return of spontaneous circulation and the theater team progressed to endoscopy.
Operative findings
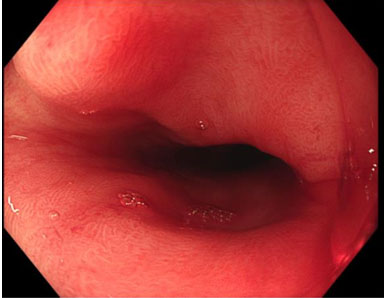
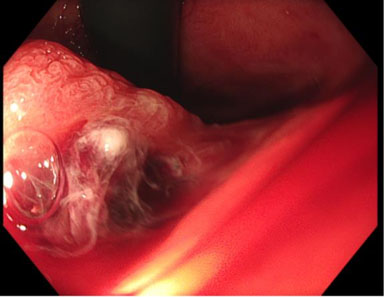
There was abundant blood in the gastric fundus (Figure 1); however, no discernible active bleeding was found in the esophagus (Figure 2), stomach, or first two parts of the duodenum. A medium-sized hiatus hernia measuring about 4 cm was noted (Figure 1) and on closer inspect a Cameron lesion was identified with overlying blood clot (Figure 3) in the hiatus hernia. Hemospray® endoscopic sealant (Cook Medical) was sprayed over the clot (Figure 4).
Post-operative course
His intraoperative arterial blood gas analysis showed a pH of 7.24 (7.30–7.40), carbon dioxide 42 mmHg (40– 50 mmHg), bicarbonate 17 mmol/L (40–50 mmHg), lactate 4.9 mmol/L (<2 mmol/L), hemoglobin 89 g/L (130–170 g/L), and ionized calcium 1.17 mmol/L (1.15– 1.30 mmol/L). Fresh frozen plasma was administered during endoscopy and a transurethral urinary catheter was inserted post-operatively. Post-resuscitation electrocardiography did not reveal any primary cardiac cause for cardiac arrest.
The patient was transferred to intensive care unit (ICU) intubated and ventilated with a proton pump inhibitor infusion and forced-air patient warming system. Chest radiography in ICU did not show any rib fractures or pneumothorax.
Community discharge
He recovered well with no further UGI bleeding and was discharged on high dose proton pump inhibitor therapy. He is booked for a surveillance gastroscopy within the next four months.
Readmission
He presented to his local hospital two months later with melena and severe anemia after non-adherence to proton pump inhibitor therapy and was transferred to a tertiary referral center [hemoglobin was 69 g/L (130–180 g/L) from 99 g/L on discharge]. Endoscopy this time revealed active bleeding from the ulcer within the hiatus hernia. Hemostasis was achieved by thermal therapy to the ulcer base. Biopsy of the ulcer could not be obtained during both procedures, given the acuity of his presentations. He has since been discharged on twice daily oral proton pump inhibitor therapy with planned surveillance gastroscopy within the next four months.
Discussion
The mechanism of Cameron lesion genesis is still debated; however, one study found them to be associated with large hiatus herniae, non-steroidal anti-inflammatory drug (NSAID) use, and increasing age [1]. They are found in 3.3–5% of patients with hiatus hernia [1],[2]. Cameron lesions can heal spontaneously and in other patients they may persist and cause iron deficiency anemia [2]. Acute upper gastrointestinal hemorrhage may occur in 36% of symptomatic patients [3]. The lesion is often missed on index endoscopy [3].
Initial treatment of upper gastrointestinal hemorrhage should include intravenous proton pump inhibitor therapy and intravenous fluid resuscitation. However, the volume of crystalloid resuscitation should be limited to an initial 10–20 mL/kg bolus which is followed by blood products guided by hematological and coagulation studies. Clot formation is promoted by increasing the pH of gastric fluid which facilitates coagulation, and a permissive hypotension strategy can be targeted if there is repeated overt hemorrhage. Tranexamic acid has been shown to decrease rebleeding rates and necessity for surgical intervention but not mortality [4].
The patient’s clinical response to medical management and resuscitation should guide the timing of upper gastrointestinal endoscopy. This report shows patients with ongoing blood loss, hemodynamic instability, or evidence of shock despite optimal medical management warrant urgent endoscopic hemostasis and/or surgery.
In Australia, most elective endoscopy procedures are performed with sedation and an unprotected airway. Clinicians should be conscious that patients with upper gastrointestinal bleeding are at high risk of airway obstruction or aspiration. Rapid sequence induction should be performed and establishing a definitive airway with a cuffed endotracheal tube placed below the level of the vocal cords is necessary. Chest compressions on patients with UGI bleeding leads to regurgitation of blood into the oropharynx which can obstruct the airway and delay or prevent endotracheal intubation and ventilation. Static blood flow promotes clot formation, hence cardiac arrest was likely the cause of no active bleeding on endoscopy despite return of spontaneous circulation. Meticulous examination of the upper gastrointestinal tract is required when no active bleeding can be found. Hiatus hernias should be closely examined for Cameron lesions in patients presenting with UGI hemorrhage.
Cameron lesions may be managed medically, surgically, and endoscopically. Medical management involves proton pump inhibitors to suppress gastric acid levels and promote mucosal healing while concurrent iron supplementation addresses iron deficiency anemia. The role of endoscopy is achieving hemostasis via band ligation, cauterization, and/or adrenaline injection; however, Cameron lesions are difficult to control endoscopically [5]. While most patients respond to medical therapy, definitive management with fundoplication is recommended for medically refractory disease, hiatus hernia incarceration, gastric volvulus or perforation [2].
Hemospray® is an endoscopic hemostatic agent developed by Cook Medical which delivers an inert, bentonite powder from a catheter passed through the endoscope [6]. Hemospray® is approved for non-variceal UGI bleeding in Australia, Europe, and Canada. Overall, 750 patients have been included over multiple clinical trials (Phase I feasibility study, SEAL survey, HALT survey, APPROACH study, Hemospray® Literature, and emergency use) showing a 95–100% success rate in achieving hemostasis and a re-bleeding rate of 10–22% [7].
Conclusion
Life-threatening upper gastrointestinal hemorrhage should be managed according to the priorities of airway, breathing, and circulation. Cameron lesions are an uncommon cause for hypovolemic shock and cardiac arrest but can be identified during upper gastrointestinal tract endoscopy; however, endoscopic hemostasis is technically difficult. Endoscopic hemostatic agents can be considered as an adjunct/alternative to mechanical agents like banding or clips. Patients with medically refractory or complicated disease should be referred for surgical fundoplication.
REFERENCES
1.
Gray DM, Kushnir V, Kalra G, et al. Cameron lesions in patients with hiatal hernias: Prevalence, presentation, and treatment outcome. Dis Esophagus 2015;28(5):448–52. [CrossRef]
[Pubmed]

2.
Brar HS, Aloysius MM, Shah NJ. Cameron Lesions. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2023.
[Pubmed]

3.
Zullo A, Manta R, De Francesco V, et al. Cameron lesions: A still overlooked diagnosis. Case report and systematic review of literature. Clin Res Hepatol Gastroenterol 2018;42(6):604–9. [CrossRef]
[Pubmed]

4.
Burke E, Harkins P, Ahmed I. Is there a role for tranexamic acid in upper GI bleeding? A systematic review and meta-analysis. Surg Res Pract 2021;2021:8876991. [CrossRef]
[Pubmed]

5.
Kapadia S, Jagroop S, Kumar A. Cameron ulcers: An atypical source for a massive upper gastrointestinal bleed. World J Gastroenterol 2012;18(35):4959–61. [CrossRef]
[Pubmed]

6.
Cook Medical. Hemospray® Endoscopic Hemostat: Instructions for use. 2023. [Available at: https://www.cookmedical.com/products/35a4a7f2-867b-4c81-a983-44ea06277852]

7.
Cook Medical. Hemospray: Summary of clinical data. 2023. [Available at: https://cookmedical.com/HemosprayData]

SUPPORTING INFORMATION
Author Contributions
Alen Brodaric - Acquisition of data, Analysis of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Rajkumar Srinivasan - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2024 Alen Maximillian Brodaric et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.