|
Case Report
A case of inflammatory breast cancer following augmentation with silicone injections
1 MD, Surgery Resident, Department of Surgery, HCA Florida Brandon Hospital/USF Morsani College of Medicine, Brandon, Florida, USA
2 MD, Surgery Professor, Department of Plastic Surgery, HCA Florida Brandon Hospital, Brandon, Florida, USA
3 MD, Surgery Program Director, Department of Vascular Surgery, HCA Florida Brandon Hospital/USF Morsani College of Medicine, Brandon, Florida, USA
Address correspondence to:
Trina Capelli
119 Oakfield Dr., Brandon, FL 33511,
USA
Message to Corresponding Author
Article ID: 100144Z12TC2024
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Capelli T, Socas J, Abbruzzese T. A case of inflammatory breast cancer following augmentation with silicone injections. J Case Rep Images Surg 2024;10(2):22–25.ABSTRACT
Inflammatory breast cancer following augmentation with silicone injections is a rare occurrence. We report a case of a 38-year-old female who presented 15 years after receiving silicone breast injections with inflammatory breast cancer. She underwent bilateral modified radical mastectomies after receiving neoadjuvant chemotherapy. The final pathology revealed invasive ductal carcinoma, ER+, PR+, HER2—with tumor cells present in the dermis of the skin. She is now undergoing radiation therapy. Breast cancer after silicone injections should be aggressively screened for with special consideration given to magnetic resonance imaging (MRI) and contrast-enhanced spectral mammography. Prompt diagnosis and treatment can improve patient outcomes.
Keywords: Breast cancer, Breast surgery, Silicone injections
Introduction
Inflammatory breast cancer following augmentation with silicone injections is a rare occurrence although the link between silicone injections and cancerous tumors has become more widely accepted. Given this acceptance, the Food and Drug Administration (FDA) banned the use of all silicone injection products in 1992 [1]. Prior to the FDA banning the use of all silicone injection products, there was widespread use of injected liquid silicone for breast augmentation. Even with silicone injections no longer legally being used for breast augmentation, women who previously have had breast augmentation via silicone injections remain at risk for silicone-associated breast cancers [2]. Often, silicone-associated breast cancers take many years to declare themselves. Diagnosis can further be delayed due to the silicone granulomas causing marked acoustic shadowing on ultrasound and decreased penetration of breast tissue via mammogram [3]. We present a case of a 38-year-old female who presented with inflammatory breast cancer 15 years after breast augmentation via silicone injections.
Case Report
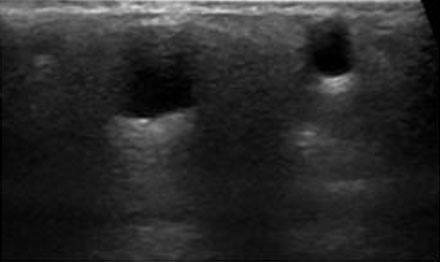
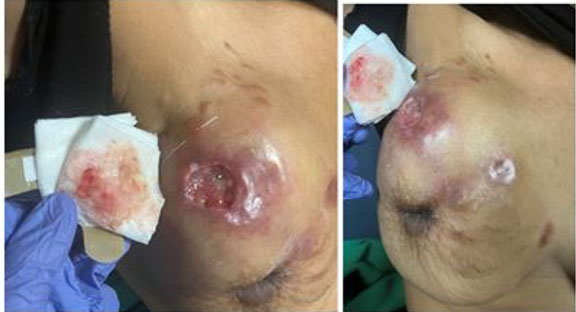
A 38-year-old female with no past medical history and a past surgical history significant for breast enhancement via silicone injections 15 years ago presented to the emergency department with complaints of right breast pain for one month. About two years prior to her presentation she had noticed what she thought was a cyst at the 12 o’clock position in her right breast. At that time, she received a mammogram and ultrasound which showed numerous well-circumscribed cysts classified as benign, BIRADS-2 (Figure 1 and Figure 2). Two months prior to her presentation, she noticed her right nipple starting to become inverted. About a month later, she developed pain at the 12 o’clock position of the right breast. Around the same time, she also noticed that she was developing erythematous nodules located along the right breast in the 2 and 3 o’clock positions. A week prior to her presentation she developed ulceration over the 12 o’clock region of her right breast with associated foul smelling drainage (Figure 3). While she denied any personal or family history of cancer, she did smoke marijuana daily.
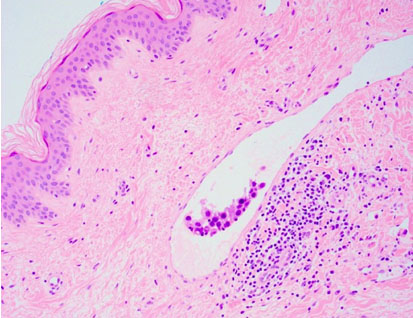
An ultrasound was performed in the emergency department which was found to be non-diagnostic with a BIRADS-0 due to the breast being so hard and dense that the ultrasound probe was unable to penetrate the tissues. She then underwent a punch biopsy of the right breast in which pathology showed invasive ductal carcinoma, ER+, PR+, HER2—with tumor cells present in the dermis of the skin, consistent with inflammatory breast cancer (Figure 4). Immunohistochemistry revealed that both her ER and PR receptors were overexpressed while her HER2 receptors were not overexpressed. Tumor cells were positive for ER and GATA-3 immunostains. D2-40 immunostain revealed few lymphatic channels without tumor. Tumor cells were present in the dermis of the skin. Pathology showed a combined histological grade of 2 (score 6/9) with tubule formation 3/3, nuclear pleomorphism 2/3, and mitotic activity 1/3. She was referred to a breast surgeon and promptly started neoadjuvant chemotherapy, which included eight cycles of Adriamycin and Taxol. She then underwent bilateral modified radical mastectomies and recovered well. She is currently undergoing radiation therapy, which consists of daily radiation dose of 50 Gray, five days a week for six weeks.
Discussion
We presented the case of a 38-year-old female with inflammatory breast cancer 15 years after undergoing breast augmentation with silicone injections. Inflammatory breast cancer is a rare form of locally advanced breast cancer accounting for only 2–4% of all breast cancer in the United States [4]. Although there is not much data regarding breast cancer arising from silicone injections, breast cancer arising after breast augmentation in general is around 0.2–2.7% [5].
The exact mechanism of how silicone injections can cause breast cancer is unknown. However, it is well known that silicone is a non-biodegradable prosthetic material that does not illicit a large local inflammatory response in most people [6]. Over the years, complications from silicone have arisen. It has been documented that, rarely, silicone can form local and systemic granulomatous inflammatory reactions affecting breast tissue along with many other organs [7]. The mechanism of this inflammatory reaction remains unknown, and a causality of silicone causing cancer remains to be proven [8].
When thinking about the association of silicone injections for breast augmentation and breast cancer, it is important to think about how silicone injections affect the ability to screen for breast cancer. It is widely accepted that the injection of silicone into the breast tissue makes it more difficult to detect cancer on routine surveillance screenings via ultrasound and mammogram [9]. Specifically, using ultrasound, silicone causes diffuse acoustic shadowing due to strong reflection and refraction of the sonographic beam [9]. This makes it very difficult to discern between the silicone and breast masses. This was seen in our case as the ultrasound was non-diagnostic due to the ultrasound wavelengths not being able to penetrate the silicone. Similarly, mammography also has a hard time distinguishing between the silicone and the breast tissue, leading to difficulty in detecting breast masses [10]. Magnetic resonance imaging (MRI) has been recommended as an additional screening tool in women with known silicone breast injections because MRI is able to discern between breast lesions (benign or malignant) and silicone [9].
Although MRI has been shown to accurately detect breast cancer in patients with a history of silicone injection, contrast-enhanced MRI biopsies often take over a month to schedule and result. This often causes a delay in diagnosis of breast cancer. Alternatively, contrast-enhanced spectral mammography (CESM) is a new technique which has shown promising results in terms of accurately detecting breast cancer in patients with silicone injected breast tissue [11],[12]. This becomes especially important when thinking about the patient population who may not be able to receive MRI, for example, those with pacemakers which are not compatible with MRI machines or even severe anxiety and claustrophobia.
Aside from screening, the idea of a prophylactic mastectomy in patients with silicone breast injections has also been proposed. While prophylactic skin/nipple sparing mastectomy has been encouraged by some, others have discouraged this as a skin/nipple sparing mastectomy that does not fully remove the injectable materials because they may have infiltrated the skin and nipple-areolar-complex. Therefore, patients can still have a chance of developing breast cancer [9]. Given the persistent risk of cancer in these patients, patient education on their options becomes increasingly important. Diligent commitment to the most accurate screening modalities may prove to be the most beneficial to patients with silicone injected breasts.
Conclusion
Breast cancer arising after silicone-injected breast augmentation is often undiagnosed until it is too late. Physicians caring for patients with silicone injection augmentation need to be aware of this potentially fatal association, especially many years after the initial injections. Physician education regarding the best screening modalities for patients with silicone breast injections is of the upmost importance. Patient education and diligent commitment to the most accurate screening modalities may prove to be the most beneficial to patients with silicone injected breasts.
REFERENCES
1.
Nakahori R, Takahashi R, Akashi M, et al. Breast carcinoma originating from a silicone granuloma: A case report. World J Surg Oncol 2015;13:72. [CrossRef]
[Pubmed]

2.
Toyonaka R, Ozeki J, Koyama Y, et al. A case of breast squamous cell carcinoma following breast augmentation with liquid silicone injection after 16 years. Surg Case Rep 2022;8(1):22. [CrossRef]
[Pubmed]

3.
Ko C, Ahn CY, Markowitz BL. Injected liquid silicone, chronic mastitis, and undetected breast cancer. Ann Plast Surg 1995;34(2):176–9. [CrossRef]
[Pubmed]

4.
Chippa V, Barazi H. Inflammatory Breast Cancer. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2024.
[Pubmed]

5.
Cheung YC, Kuo WL, Lee LY, Tang YC. A case report of breast cancer in silicone-injected breasts diagnosed by an emerging technique of contrast-enhanced mammography-guided biopsy. Front Oncol 2022:12:884576. [CrossRef]
[Pubmed]

6.
Adams ST, Cox J, Rao GS. Axillary silicone lymphadenopathy presenting with a lump and altered sensation in the breast: A case report. J Med Case Rep 2009;3:6442. [CrossRef]
[Pubmed]

7.
Cook PD, Osborne BM, Connor RL, Strauss JF. Follicular lymphoma adjacent to foreign body granulomatous inflammation and fibrosis surrounding silicone breast prosthesis. Am J Surg Pathol 1995;19(6):712–7. [CrossRef]
[Pubmed]

8.
Tugwell P, Wells G, Peterson J, et al. Do silicone breast implants cause rheumatologic disorders? A systematic review for a court-appointed national science panel. Arthritis Rheum 2001;44(11):2477–84. [CrossRef]
[Pubmed]

9.
Prasetyono TOH, Sadikin PM. Management of asymptomatic silicone-injected breast with reduction mammoplasty. Indian J Plast Surg 2015;48(3):317–20. [CrossRef]
[Pubmed]

10.
Institute of Medicine (US) Committee on the Safety of Silicone Breast Implants. Safety of Silicone Breast Implants. Bondurant S, Ernster V, Herdman R, editors. Washington (DC): National Academies Press (US); 1999. [CrossRef]
[Pubmed]

11.
Hogan MP, Amir T, Mango VL, Morris EA, Jochelson MS. Feasibility of contrast-enhanced mammography in women with breast implants. Clin Imaging 2023;93:31–3. [CrossRef]
[Pubmed]

12.
Neelamraju Lakshmi H, Saini D, Om P, Verma N. A case of carcinosarcoma of the breast presenting as inflammatory carcinoma and review of the literature. Cureus 2020;12(8):e10104. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Trina Capelli - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Juan Socas - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Thomas Abbruzzese - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2024 Trina Capelli et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.